Due to the increased awareness of food hygiene, the European Hygienic Engineering and Design Group (EHEDG) was founded.
The EHEDG objective is to improve product safety from a scientific and technological point of view and from the EC legislation on the hygienic design of food machines.
This article will discuss:
- Hygienic design guidelines
- Test procedure
- Cleaning procedure test section
- Detection of residual ground
- Cleaning assessment and interpretation
The experts at Dinnissen Process Technology are available to answer all your questions:
Get in touch with Juul Jenneskens 077 467 3555
Hygienic design guidelines
EHEDG has established hygienic design guidelines that can be verified by standard test procedures related to hygienic design. This includes cleanability, pasteurizability, serialisability and aseptic capability.
The test procedure is a basic screening test to identify areas of poor hygienic design and where product or microorganisms are protected from the action of the cleaning process. It can also be used to compare appliances with regard to their cleanability in place. The test is not indicative of performance in industrial cleaning situations.
The cleanliness grade is based on the removal of a soil containing bacteria and is assessed by evaluating the growth of residual bacteria after cleaning. The following terms are used for this:
- Cleaning: the removal of soil, food residues, dirt, grease or other annoying substances,
- Cleanability: the suitability of equipment to be easily freed from soil,
- Comparative cleanability: the cleanability of equipment in relation to a reference,
- On-site cleanability: the ability to be cleaned easily without disassembly.

EHEDG
Test procedure
First, a test section must be formed. This is done by connecting the equipment to be tested to a 250 mm reference pipe. With the known internal surface roughness (mean Ra = 0.5 - 0.7 microns or equivalent according to ISO 4287: 1977) and with two short auxiliary lengths made of pipe at each end (Fig. 1) to form a test section.
After this, the test part is filled with sour milk and both ends are closed. After this, it is pressurised three times to 5 bar, using a closing plate with a suitable air coupling, for two minutes. It is possible to stimulate a commissioning situation of the test section by operating all movable parts. A total of 10 surgeries must be performed within the 3 pressurised periods.
Then the sour milk is poured off and the test portion dried by rinsing with dry filtered air.
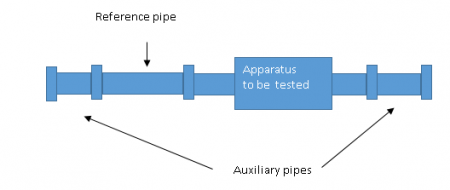
Test procedure
Test section cleaning procedure
For cleaning, the dirty test area is mounted in a specially built test setup, without removing the auxiliary pipes. A straight tube, at least five times the diameter of the reference pipe, must be inserted before and after the test section to ensure a stabilized flow of cleaning solution through the test section. Then the following cleaning procedure can begin:
- Rinse with cold water (10-200C) for a time equal to the mean residence time (t) and not less than 1 minute,
- Circulate a 1% (w / v) detergent solution at 630C +/- 20C for 10 minutes (the volume of the detergent solution used must be at least 20 times the internal volume of the test item),
- Rinse with cold water (10-200C) for a time equal to the mean residence time (t) and not less than 1 minute.
- Where mean residence time (t) = volume of test item / flow rate.
At the outlet of the equipment, water samples are taken as accurately as possible from both flushes at the end of the flushing procedures. Two 5 ml samples are poured with MSHA per petri dish. Pre-rinse will recover 3-300 spores per 10 ml. After the final rinse, <1 spore per 10 ml should be recovered. Less than 1 spore per 10 ml in the pre-rinse or the presence of spores in the final rinse may indicate an inadequate test procedure and could lead to false results.
Detection of residual earth
After cleaning, the test portion is removed from the rig. The inner surface of the reference pipe is covered with molten MSHA to a thickness of at least 5 mm, including all relevant surfaces of the test equipment. After the agar has completely set, the reference pipe is placed almost vertically in a 580 incubator for 24 hours. Condensation in the reference pipe may spread microorganisms and / or acidic microbial metabolic products along the reference pipe during incubation. In this situation a false reading of yellow discoloration is obtained.
The test procedure is a basic screening test to identify areas of poor hygienic design and where product or microorganisms are protected from the action of the cleaning process
Cleaning assessment and interpretation
After incubation, the test equipment and reference pipe are examined for the presence of yellow discoloration of the purple MSHA. It is recommended to use a 5x5 mm scale grid and a color comparison disc to standardize the test results of different operators.
The disc contains graduated shades of yellow to purple. The transition point to count as yellow is shade 3, shade 4 is light purple. Indicative of a normal reference pipe cleaning procedure is a small yellow area (approx. 5-30%). If the yellow area is greater than 30%, the test must be repeated as interpretation of these results is difficult.
In general, three results are possible, assuming there is a small amount of yellow in the reference pipe:
- Presence of milk residues,
- Presence of yellow zones and / or colonies,
- No yellow zones / colonies.
If visible milk residues are observed before the agar is applied when disassembling the test object, no microbiological examination is necessary as traces are clearly present. However, a microbiological analysis of the reference pipe is required to confirm that the cleaning was acceptable. There may be a hygienic design flaw inherent in the test item or a flaw in the test methodology. After this, the test must be repeated. If milk residues are found again in the same area, design changes must be made by the test item manufacturer. If the test method was incorrect, it must be corrected and the test repeated.
Test procedures should be repeated up to five times to investigate whether yellow areas in the test item are randomly distributed or indicate poor hygienic design. If there is three soil remaining in the same area of the test specimen, it indicates areas that are difficult to clean. Improvements in hygienic design should be considered.
If the percentage of yellow zones in the test item is comparable to that of the reference pipe, then the degree of cleanability is also comparable. If the percentage of yellow zones in the test item is smaller or larger than the reference pipe, the test item is more or less cleanable.
In some cases, there may be no yellow zones left in the test item. If this is on three consecutive occasions, no further test repetitions are required, the test item can be described as particularly cleanable.
Occasionally, some gasket materials will give false negative test results in areas of poor hygienic design, while there are no obvious yellow areas. This is possible because the packing material has antibacterial properties, so that spores present on their surface are prevented from germinating and / or growing.
All o-rings and gaskets in the test item should be checked for antibacterial properties. The gasket / o-rings can have antibacterial properties if no yellow discoloration is visible in the agar directly around the sample. The test should be performed with gaskets / O-rings of a different material until yellow discoloration is visible in the agar.
Gaskets / o-ring with antibacterial properties show a purple zone around them. A different material should be used to perform the test until no purple discoloration is seen in the yellow agar. The gaskets / o-rings have no antibacterial properties if the agar in the petri dishes is completely yellow.

Name: Juul Jenneskens
Advisor
Please feel free to contact me if you have any questions about this subject. My team of colleagues and I are ready to answer!
Get in touch with Juul Jenneskens 077 467 3555 [email protected]
Do you prefer to request a consultation directly?
